Making a run to the home improvement store for your next project? That’s what scientists in the United States did when they needed red bricks – the common kind used in buildings across the world – to prove their theory that the bricks could be transformed into a kind of battery to store energy used in homes and offices.
Julio D’Arcy, an assistant professor of chemistry at Washington University in St Louis, said he paid 65 cents each for bricks at Home Depot, a familiar North American home improvement retail chain, while underscoring just how readily available is a basic material that can be converted into “smart bricks” once it is treated with a polymer coating.
“Our method works with regular brick or recycled bricks, and we can make our own bricks as well,” said D’Arcy. His team, led by graduate student Hongmin Wang, published their work last week in the journal Nature Communications.
The scientists in St. Louis developed a coating of PEDOT, a polymer made of nanofibers that is absorbed into the fired brick and stays trapped in the porous material. PEDOT, known chemically as poly(3,4-ethylenedioxythiophene), has electronic conductivity properties that can convert the brick into a kind of supercapacitor.
The process also relies on the red pigment in bricks – iron oxide, or rust – to trigger the polymerization reaction. The brick then functions like an ion sponge that can store energy like batteries do. In the above illustration, provided by D’Arcy’s lab, the green LED light is powered directly by the brick.
“PEDOT-coated bricks are ideal building blocks that can provide power to emergency lighting,” D’Arcy said. “We envision that this could be a reality when you connect our bricks with solar cells — this could take 50 bricks in close proximity to the load. These 50 bricks would enable powering emergency lighting for five hours.”
A brick wall serving as a supercapacitor can be recharged hundreds of thousands of times within an hour, he adds, noting that “if you connect a couple of bricks, microelectronics sensors would be easily powered.”
The proof-of-concept research published last week builds on work the Washington University team has done with rust to develop its potential as a supercapacitor. They’ve been using specially designed instruments to work with the rust, chemically known as Fe2O3, to deliver tiny layers of metal oxide film as thin as 20 nanometers.
“The easiest way to remove rust from a surface is to add a little bit of acid. That’s what the rust remover you get at the hardware store is,” says D’Arcy. “Our conversion works the same way – we add acid and change the iron oxide by liberating an iron atom. That iron atom becomes the reactant for our polymer vapor.”
D’Arcy’s team is aware that research on red brick has demonstrated its potential in absorbing heat, but they say this is the first time that a “dirt-cheap” construction material might be used to provide energy storage for microdevices that are a part of the building itself.
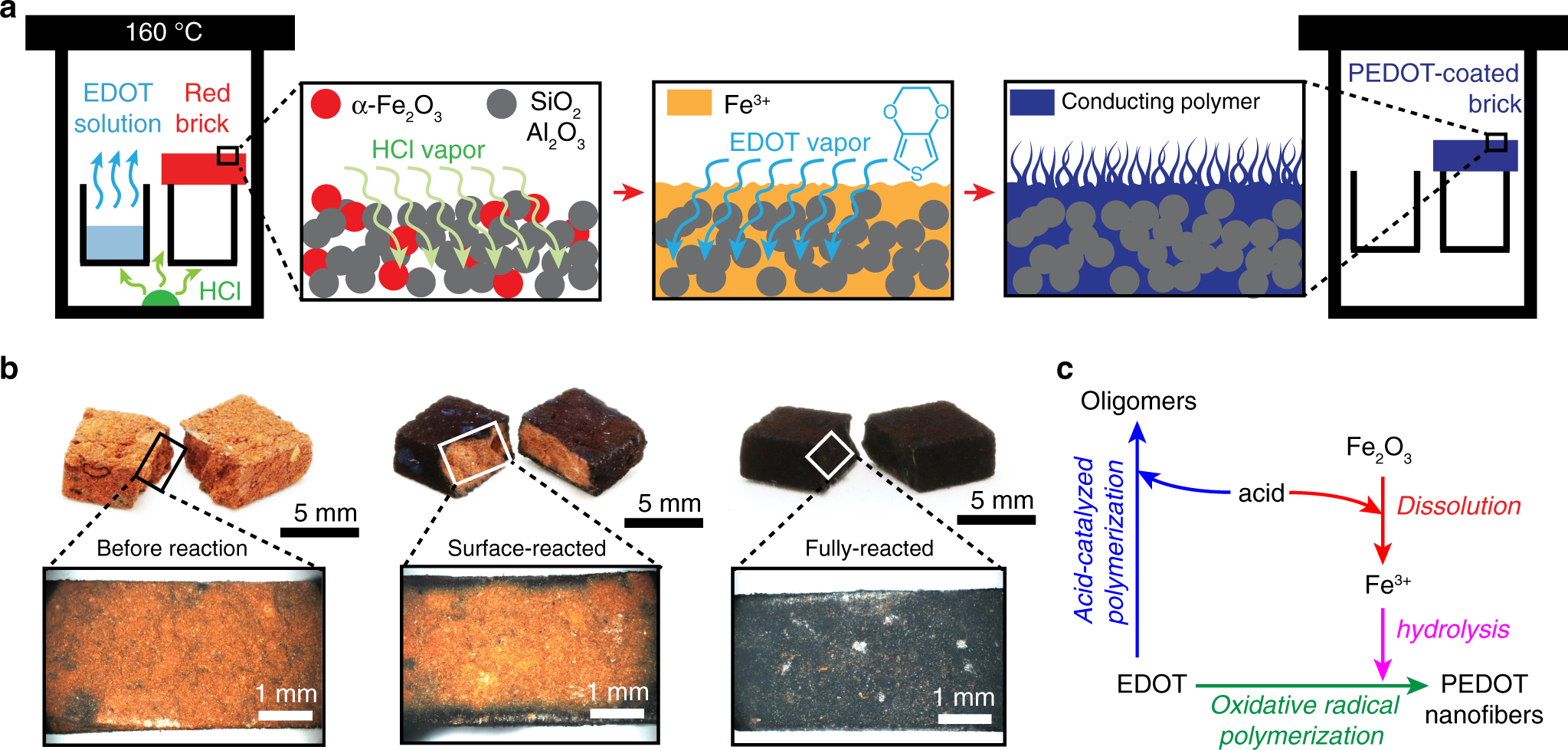
a In a one-step reaction, a brick’s α-Fe2O3 microstructure is partially dissolved by acid vapor to liberate Fe3+, promote hydrolysis and precipitation of FeOOH spindles that control oxidative radical polymerization. As previously reported, monomer vapor reacts with partially dissolved FeOOH nuclei resulting in preferential directional growth of high aspect ratio PEDOT nanofibers13. b The thickness of a PEDOT coating is controlled by reaction time and stoichiometry enabling a reaction to generate surface-polymerized PEDOT-coated bricks (core/shell architecture) or fully polymerized bricks (monolithic PEDOT architecture). c A reaction diagram shows the competition between acid-catalyzed polymerization and oxidative radical polymerization mechanisms present in our reactions.




